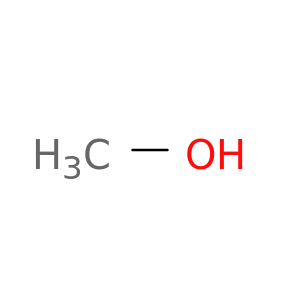
Methanol
Molecule Viewer
Thermodynamics at 298.15 K and standard pressure
| Enthalpy | ||
| Property | Value | Unit |
| Translational | 6.20 | kJ mol-1 |
| Rotational | 3.72 | kJ mol-1 |
| Vibrational | 133.71 | kJ mol-1 |
| Total (Trans. + Rot. + Vib.) | 143.63 | kJ mol-1 |
| Heat Capacity at Constant Pressure | ||
| Property | Value | Unit |
| Translational | 20.79 | J mol-1 K-1 |
| Rotational | 12.47 | J mol-1 K-1 |
| Vibrational | 13.03 | J mol-1 K-1 |
| Total (Trans. + Rot. + Vib.) | 46.29 | J mol-1 K-1 |
| Entropy | ||
| Property | Value | Unit |
| Translational | 151.98 | J mol-1 K-1 |
| Rotational | 79.18 | J mol-1 K-1 |
| Vibrational | 7.30 | J mol-1 K-1 |
| Total (Trans. + Rot. + Vib.) | 238.46 | J mol-1 K-1 |
| Other Properties | ||
| Property | Value | Unit |
| Heat of Formation | -216.85 | kJ mol-1 |