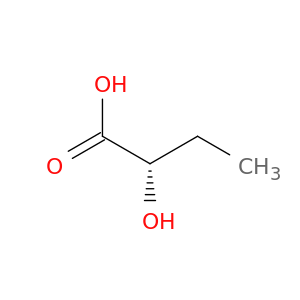
(2s)-2-hydroxybutanoic acid
Molecule Viewer
Thermodynamics at 298.15 K and standard pressure
| Enthalpy | ||
| Property | Value | Unit |
| Translational | 6.20 | kJ mol-1 |
| Rotational | 3.72 | kJ mol-1 |
| Vibrational | 335.97 | kJ mol-1 |
| Total (Trans. + Rot. + Vib.) | 345.88 | kJ mol-1 |
| Heat Capacity at Constant Pressure | ||
| Property | Value | Unit |
| Translational | 20.79 | J mol-1 K-1 |
| Rotational | 12.47 | J mol-1 K-1 |
| Vibrational | 95.03 | J mol-1 K-1 |
| Total (Trans. + Rot. + Vib.) | 128.29 | J mol-1 K-1 |
| Entropy | ||
| Property | Value | Unit |
| Translational | 166.67 | J mol-1 K-1 |
| Rotational | 116.12 | J mol-1 K-1 |
| Vibrational | 91.15 | J mol-1 K-1 |
| Total (Trans. + Rot. + Vib.) | 373.94 | J mol-1 K-1 |
| Other Properties | ||
| Property | Value | Unit |
| Heat of Formation | -624.87 | kJ mol-1 |
Molecular Orbitals
- 1 -552.31 eV View
- 2 -551.92 eV View
- 3 -550.87 eV View
- 4 -303.70 eV View
- 5 -302.82 eV View
- 6 -301.26 eV View
- 7 -300.75 eV View
- 8 -37.69 eV View
- 9 -36.12 eV View
- 10 -34.49 eV View
- 11 -28.12 eV View
- 12 -25.03 eV View
- 13 -21.96 eV View
- 14 -19.94 eV View
- 15 -18.55 eV View
- 16 -16.90 eV View
- 17 -16.81 eV View
- 18 -15.28 eV View
- 19 -14.44 eV View
- 20 -14.12 eV View
- 21 -13.71 eV View
- 22 -13.55 eV View
- 23 -12.61 eV View
- 24 -12.35 eV View
- 25 -11.56 eV View
- 26 -11.12 eV View
- 27 -9.17 eV View
- 28 -8.64 eV View
- 29 8.45 eV View
- 30 14.06 eV View
- 31 15.32 eV View
- 32 16.19 eV View
- 33 17.10 eV View
- 34 17.16 eV View
- 35 17.96 eV View
- 36 18.74 eV View
- 37 18.93 eV View
- 38 20.11 eV View
- 39 20.37 eV View
- 40 20.66 eV View
- 41 21.00 eV View
- 42 23.03 eV View
- 43 27.66 eV View
Vibrational Frequencies
| Vibration |
|
| Vectors |
|
| Balls |
|
- 1 33.00 cm-1 View
- 2 83.20 cm-1 View
- 3 186.35 cm-1 View
- 4 208.06 cm-1 View
- 5 286.16 cm-1 View
- 6 320.03 cm-1 View
- 7 365.93 cm-1 View
- 8 444.02 cm-1 View
- 9 468.51 cm-1 View
- 10 478.77 cm-1 View
- 11 558.46 cm-1 View
- 12 693.58 cm-1 View
- 13 827.22 cm-1 View
- 14 883.66 cm-1 View
- 15 971.01 cm-1 View
- 16 1035.59 cm-1 View
- 17 1052.43 cm-1 View
- 18 1115.07 cm-1 View
- 19 1127.25 cm-1 View
- 20 1138.73 cm-1 View
- 21 1175.87 cm-1 View
- 22 1295.09 cm-1 View
- 23 1324.38 cm-1 View
- 24 1328.52 cm-1 View
- 25 1388.13 cm-1 View
- 26 1394.90 cm-1 View
- 27 1406.35 cm-1 View
- 28 1408.62 cm-1 View
- 29 1423.15 cm-1 View
- 30 1428.35 cm-1 View
- 31 1982.19 cm-1 View
- 32 2891.84 cm-1 View
- 33 2953.62 cm-1 View
- 34 3033.02 cm-1 View
- 35 3079.62 cm-1 View
- 36 3084.82 cm-1 View
- 37 3181.01 cm-1 View
- 38 3869.43 cm-1 View
- 39 3874.77 cm-1 View
Aqueous Solvation Energy at 298.15 K
| Surface |
|
| Dipole |
|
| Translucent Molecule |
|
| Property | Value | Unit |
| Total Solvation Energy | -15.59 | kJ mol-1 |
| Polar Solvation Energy | -31.22 | kJ mol-1 |
| Nonpolar Solvation Energy | 15.63 | kJ mol-1 |
| Surface Area | 106.80 | Å2 |
| Charge of Molecule | 0 | |
| Dipole | 4.20 | Debye |